Clinical Trials
- Interventional/experimental trials
- Usually include randomisation
- They are prospective (individuals are followed forward from some point in time)
- Causal analysis – test causal relationship between exposure of interest & outcome
Phases of clinical trials
- Translational pathway of clinical trials:
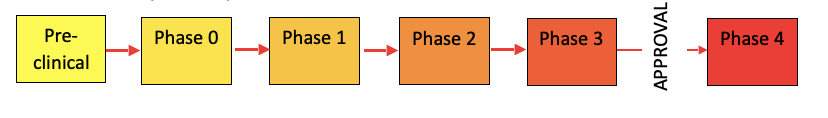
- Pre-clinical trials:
- Before testing in humans
- Helps decide whether the drug is ready for clinical trials (from ‘bench to bedside’)
- Look at toxicity, pharmacokinetic and safety information
- Typically testing in vivo (laboratory animals) or in vitro (test tubes and cell cultures)
- Phase 0
- First in-human trials
- Microdosing studies at subtherapeutic levels
- Very small studies
- Look at pharmacokinetics & pharmacodynamics – not establishing response
- All active interventions
- Not randomised/blinded
- Phase 1
- Aims:
- Establishing dose limiting toxicity
- Identify recommended phase II dose
- Also: toxicity profile, pharmacokinetics/pharmacodynamics
- All active interventions
- Not randomised/blinded
- Phase 1 starting dose options:
- 1/10th LD10 in rodents
- 1/5th TDL in rodents
- 1/3rd TLD in larger animals
- LD10: lethal dose in 10% animals
- TLD: toxic low dose – lowest dose to cause animal toxicity
- MTD: maximum tolerated dose – dose with DLT in pre-determined proportion of pts (33%)
- Recommended phase II starting dose is 1 level lower than MTD
- Aims:
- Phase 2
- Establishing potential biological effect
- To aim to exclude inactive drugs from further development – although phase 2 is NOT a definitive test of clinical efficacy
- Uses larger sample sizes with more inclusion/exclusion criteria
- Classical phase 2 trials have a single arm with no in-built controls
- Phase 3 (usually randomised controlled trial)
- Establishing efficacy (and safety)
- Larger sample sizes
- Longer follow up
- Usually blinded
- Regulatory approval (MHRA) based on these trials
- Phase 4
- Post marketing studies once drug is already in use
- Focus is to assess safety (and efficacy & cost effectiveness) in a more generalised population
- Largest sample sizes and longest follow up
- Usually observational
Design elements of clinical trials
- Trial designs
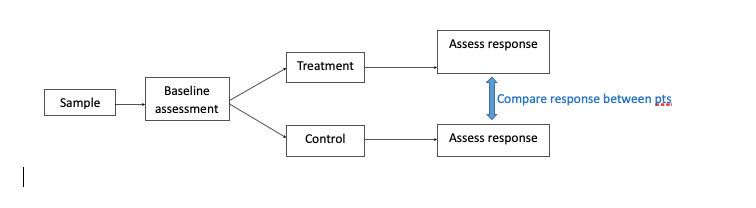
- Classic/parallel design
- ≥2 groups with follow up –> compare response between patients
- Participants receive 1 intervention each
- Most common design
- Classic/parallel design
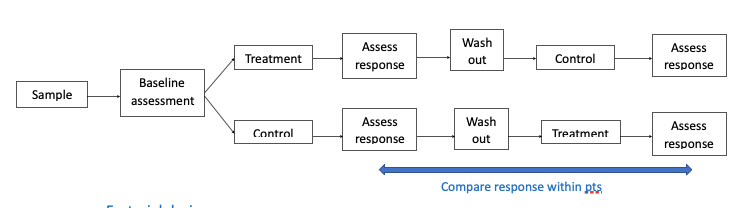
- Crossover design
- Each participant receives both interventions —> compare response within patients
- Advantages – participants are their own control so influence of confounding variables is reduced, can have smaller sample sizes
- Disadvantages – crossover effect (unclear what to do if carry-over effect are found), order effect (could order of drugs affect outcome)
- Factorial design
- Multiple interventions e.g. A, B, A&B, nothing
- Allows simultaneous analysis of a number of factors of interest e.g. adherence trial
- An efficient method of evaluating more than one intervention providing the absence of interactions
- Basket trial – targeted therapy in diseases with common pathway – MULTIPLE DISEASES 1 TREATMENT
- Umbrella trial – disease with multiple targeted therapies – MULTIPLE TREATMENTS 1 DISEASE SITE
- Platform trial – multiarm multistage
- Randomisation
- Randomisation is the process of dividing participants into groups randomly with no predetermined factors influencing this apart from chance
- Allocation ratio is usually 1:1, which usually has the most power (not always)
- Types:
- Simple
- Examples – random numbers or computer generated randomisation
- Each allocation is independent of the others
- This does risk imbalanced groups
- Block
- Relatively small block sizes are chosen, then patients are allocated to each block
- Ensures roughly equal sized treatment groups
- Fixed block size = constant block size – needs to be hidden from team (otherwise this method can be manipulated by the researcher who knows which block the patient may be allocated)
- Random block sizes = varied block sizes
- Cluster
- Randomisation of ‘clusters’ of patients rather than randomisation of individuals
- Example – Randomisation of hospital sites
- Stratified
- If there are factors which may alter outcome – these could be used to form stratum to control for these effect of these factors
- Used with cluster or block randomisation
- Example – block randomisation which has been stratified for grade of breast cancer…
- Simple
| G1 | G2 | G3 | ||||||||
| Block | Patient ID | Allocation | Block | Patient ID | Allocation | Block | Patient ID | Allocation | ||
| 1 | 1234 | A | 1 | 7890 | B | 1 | 5567 | A | ||
| 1 | 2345 | B | 1 | 8901 | B | 1 | 6678 | B | ||
| 1 | 3456 | B | 1 | 9012 | B | 1 | 7789 | A | ||
| 1 | 4567 | B | 1 | 2234 | A | 1 | 8890 | A | ||
| 2 | 5678 | A | 2 | 3345 | A | 2 | 9901 | B | ||
| 2 | 6789 | A | 2 | 4456 | A | 2 | 0012 | B | ||
| … | … | … | … | … | … | … | … | … |
- Stratified randomisation vs minimisation
- Both achieve the same thing in balancing factors between groups
- Stratified randomisation is fixed into stratum
- Minimisation is based on statistical algorithm which can vary during course of recruitment depending on which patient is recruited into which group
- Blinding
- Blinding/masking is when patients/clinicians/researchers are unaware of treatment allocation
- Reduces assessment bias
- Can be before (allocation concealment) & after intervention (blinding)
- Single = participant
- Double = participant, research team
- Triple (rarely) = participant, research team, statistician
Ethics
- GCP – trial protocols & ethics
- Trial protocol
- Defines how the clinic trial will be run (contents can vary as it is not officially regulated)
- Produced before trial is commenced
- Usually contains:
- Title & registration number
- Chief investigator details & trial team
- Version of protocol & date (to know whether it has been updated or not)
- Funding
- Roles & responsibilities of those involved in trial
- Background, objectives of the trial, patient selection criteria & methods
- Trial design, treatment & clinical evaluation
- Ethics & ethics approval
- Ethics
- Prior to 1947 there were no ethical principles to guide undertaking of clinical research
- Nuremburg code – introduced to avoid unnecessary or harmful trials without patient consent
- Followed by Declaration of Helsinki (1964) – which outlines moral principles – the responsible investigator should ensure that the study is conducted in agreement with the Declaration of Helsinki
- Led to International Council for Harmonisation Good Clinical Practice Guideline
- Informed consent
- Patients need to be informed of the details of the study including: aims; procedures; possible adverse effects & mechanism of treatment allocations
- Patients can decline further participation at any point in the trial
- The informed consent procedure must conform with GCP